Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
Incredible microscope images of molecules show what Titan's haze looks like up *very* close

Titan is the largest moon of Saturn, and the second largest moon in the solar system, about the same size as Mercury. Unique among moons, it has a thick atmosphere — despite the lower gravity, the surface pressure is 1.5 times Earth's at sea level.
Its atmosphere is 95% nitrogen (Earth's is 78%) and 5% methane. Normally that would be transparent, but Titan's air is loaded with haze — tiny particles about a micron across (one-millionth of a meter; a human hair is roughly 50–100 microns wide). These particles are suspended in the atmosphere, making it opaque.
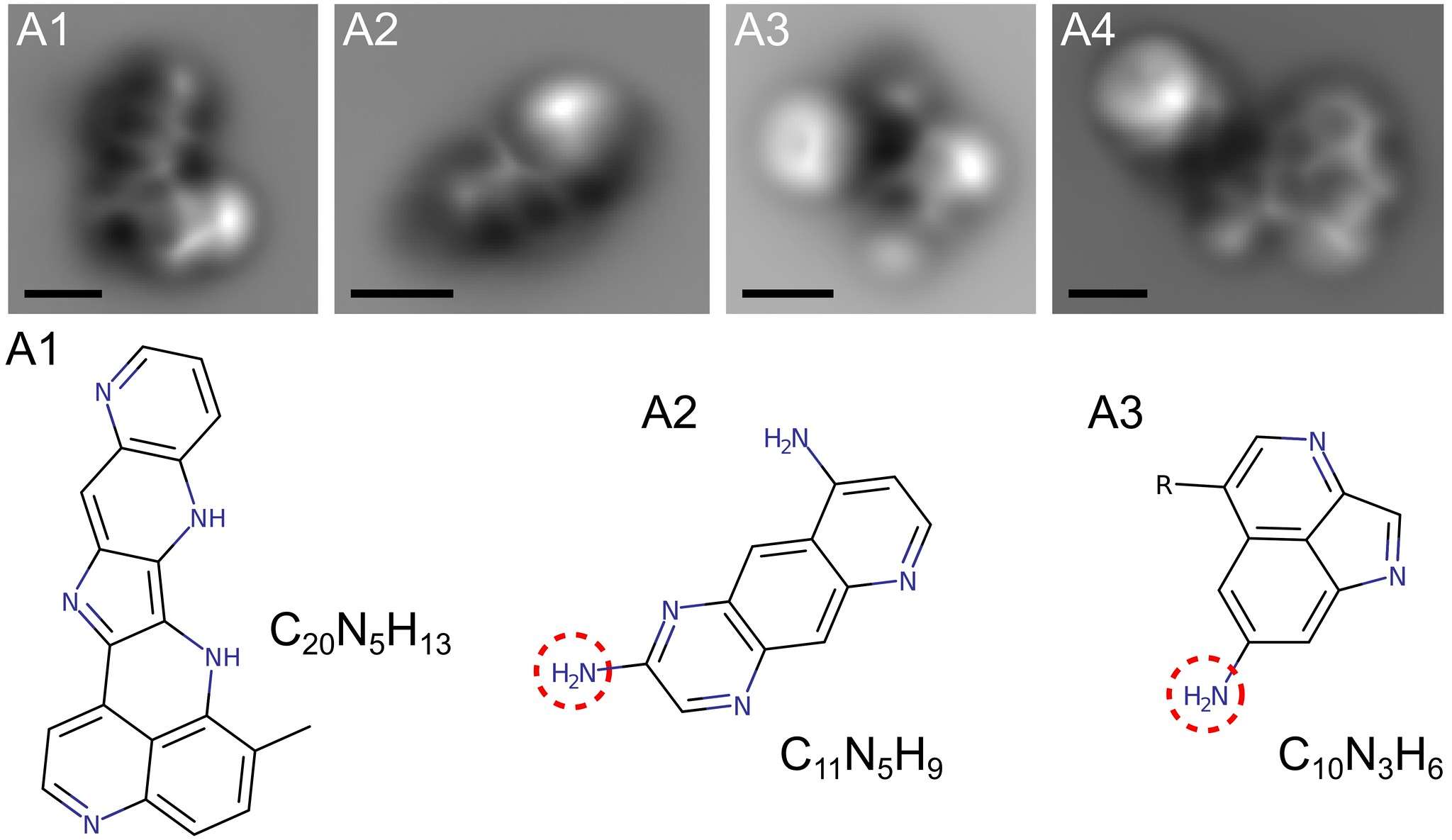
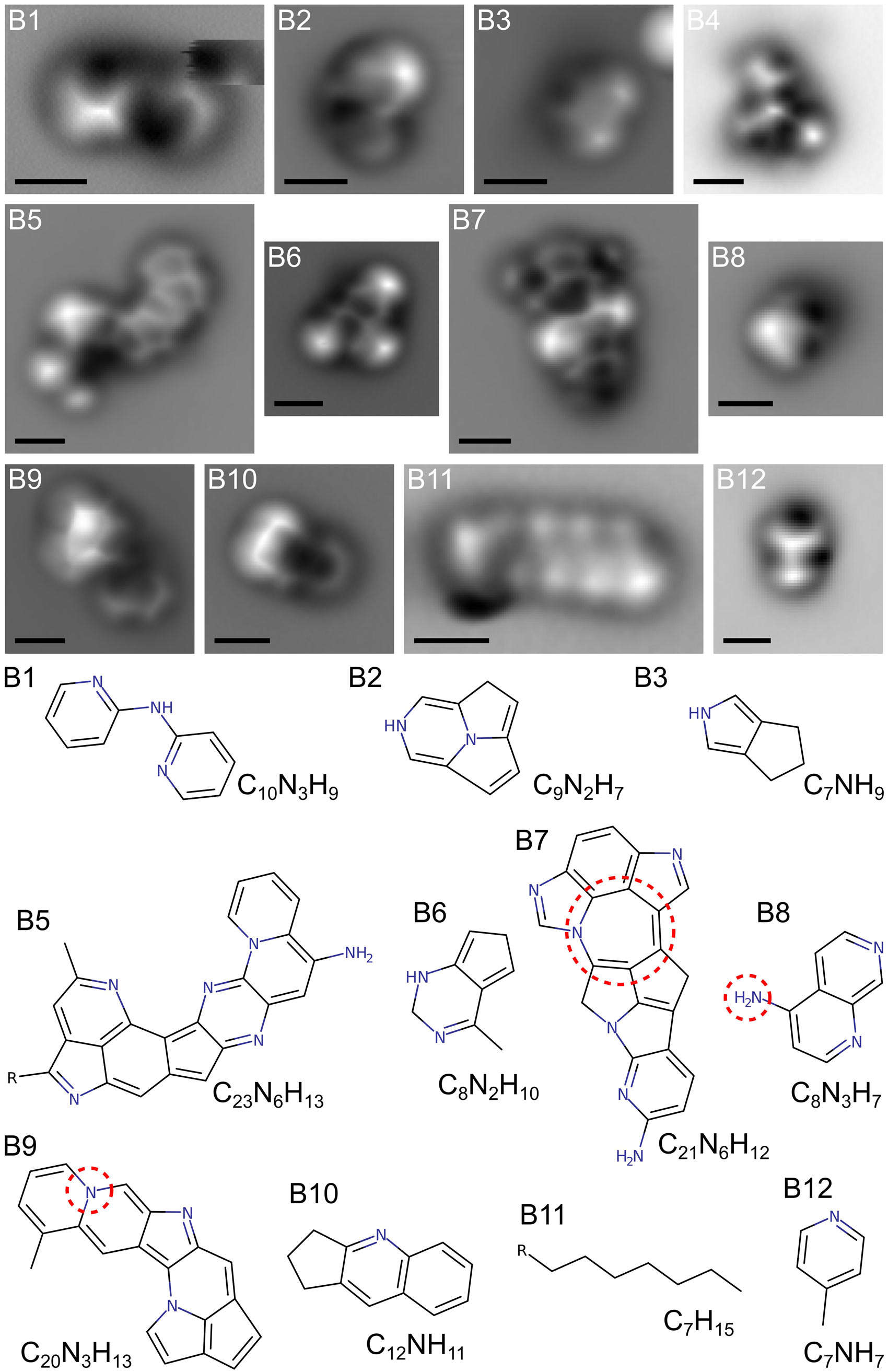
The haze particles are formed when ultraviolet light from the Sun and/or subatomic particles zipping around space slam into the nitrogen and methane, breaking it down into elements that then rearrange themselves into more complex molecules. Some of them are simple rings of carbon, and some are far more complex molecules called PAHs — polycyclic aromatic hydrocarbons. It's not been clear how the simple ones link up to form the bigger ones, but now, for the first time, this process has been simulated in a lab and the results examined using a powerful type of microscope that reveals the basic atomic configurations of the molecules.
That's amazing. Those are individual molecules you're seeing in those images. The scalebar is 0.5 nanometers, half of a billionth of a meter. They're not images like a photograph, though. It's literally impossible to do this with visible light; the wavelength of light is hundreds of nanometers, too long to see structures this small. Instead, they used what's called atomic force microscopy*.
This uses a technique analogous to the way phonographs work, by using a needle at the end of an arm that traces the grooves in a record. In this case though, a molecule at the tip of a microscopic needle runs along a molecule and can detect the change in the shape due to atomic forces holding the molecule together. It's like running your fingers over an object to feel its shape.
The samples of molecules were created in a lab to simulate Titan's atmosphere. Scientists filled a stainless steel vessel with a gaseous mixture that's the same as Titan's air and used an electric discharge (a spark maker, essentially) to simulate the UV and cosmic rays hitting the gas. It's not exactly like Titan: They did this at room temperature, which is much warmer than Titan, but the reactions aren't very sensitive to temperature. They also used a gas pressure of about 0.001 Earth's, which, though very thin, is much higher than the top of Titan's atmosphere where the reactions take place. However, the higher pressure allows the reaction rate to be much higher, so they aren't waiting weeks to get a decent sample.
They found over a hundred different molecules, a dozen or so of which they could examine using their microscope. Many are simple carbon rings and more complex PAHs, as expected. But they also found that many of the PAHs had a nitrogen atom embedded in them, making what are called N-PAHs. These molecules were detected in Titan's atmosphere by the Cassini mission, which orbited Saturn for 13 years and made over 100 passes of Titan during that time, examining its surface and atmosphere. The simulations in the lab confirm this result.
Moreover, the lab experiment created molecules made of many connected rings, up to seven of them, which will help atmospheric scientists understand how the more complex PAHs are made from simpler molecules.
This work is important for many reasons. Titan's atmosphere is loaded with this stuff, collectively called tholins (Greek for “mud”, since they make molecules which color the environment yellow, orange, and reddish-brown), and they're also seen on other worlds; Pluto's reddish colored landscape is due to tholins.
Titan doesn't have a water cycle like Earth, but it does have a methane cycle: Liquid methane in vast lakes at its north pole evaporates into the atmosphere, rains down on the hills nearby, then flows back into the lakes. Methane vapor may condense on the suspended tholins, helping it rain out, and then the tholins can coat the moon's surface. That's very interesting, because nitrogen and carbon molecules are important in prebiotic chemistry, making up amino acids, which in turn are the building blocks of proteins.
Earth's early atmosphere was likely very similar to Titan's, before the Great Oxygenation Event about 3 billion years ago which gave us the atmosphere, more or less, we have today. Studying Titan is like studying ancient Earth. Not to be too broad, but life evolved on Earth in that early atmosphere, so it's not too silly to wonder if something similar is occurring on Titan. We certainly don't know if life is brewing or thriving there, but it's certainly within the realm of science to look into it.
Titan is an alien world over a billion kilometers from the Sun, and drier than any desert on our own planet. Yet there are aching similarities, ones we can study in the lab. NASA is already in the early stages of planning a mission to Titan called Dragonfly — a lander and quadcopter drone that will fly over the surface and examine regions likely to have or have had conditions conducive to life.
What will it find there? These lab results are an important step in figuring that out.
*Just typing those words makes me feel like a scientist in an old black-and-white sci-fi movie.