Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
Could a worm with no brain help us reverse brain damage?
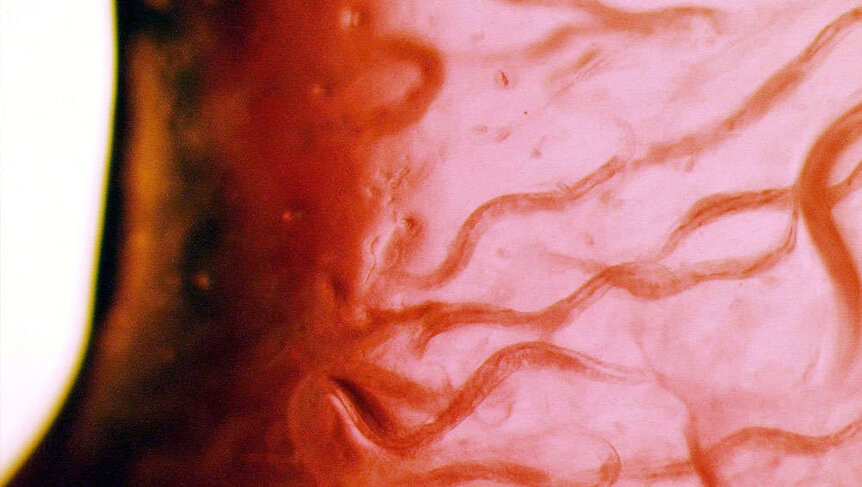
Nematodes are worms without a brain, so it might seem impossible for these lowly creatures to help repair human brains someday, but they do have one thing we have — neurons.
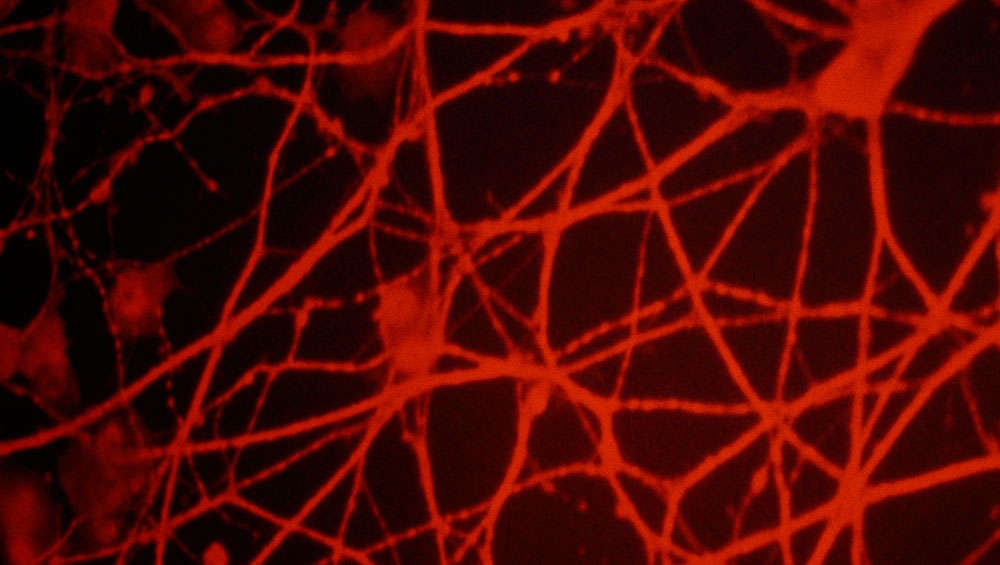
Genetic material from the brainless nematode C. elegans to repair a broken neuronal pathway and enable them to smell again. The neurons throughout these worms’ bodies zap messages back and forth. Just like in the human brain, if a message hits a dead zone, the message will fail to send, much like an email when your internet is down. Neurobiologist Ithai Rabinowitch, of Hebrew University in Israel, and colleague Jihong Bai, of the Fred Hutchinson Research Center in Seattle, restored the worms’ sense of smell by creating an artificial pathway between two neurons. This could have huge implications for the future of treating human brain disease and trauma.
“it is quite remarkable how much similarity does exist between nematodes and humans, especially at the level of the molecular components that drive the basic operation of neurons, including ion channels that regulate neuronal electrical activity, and synaptic proteins that underlie the transmission of information between neurons," Rabinowitch, who co-led a study recently published in Cell Systems, told SYFY WIRE.
C. elegans are not parasites (unlike some nematodes that can feast on you from the inside). They might be primitive compared to us, but they still experience much of our biological phenomena on a cellular level. They mate by fertilizing eggs with sperm. Their cells split and develop in the egg much like ours do in the womb. They gradually age until death. While they don’t have the type of brain that zombies are ravenous for, what they do have is a network of neurons throughout their bodies along with something called a circumpharyngeal nerve ring, or a bunch of nerves in their throats, which is as close to a brain as they can get. These tiny worms that are no more than a millimeter long are even capable of learning and exhibiting certain behaviors.
The nerve ring is obviously nowhere near the level what our brains are capable of. There is some organization, and though it still can't compare to the information superhighway which the human brain evolved into, there were still similarilties that Rabinowitch was able to use to his advantage when experimenting on the nematodes.
"The C. elegans nerve ring and the human brain both serve as the major junction for neuronal communication and interaction through synaptic transmission," he said. "Unlike the human brain, the nerve ring does not contain neuron cell bodies, only their processes (the cell bodies are clustered in various areas of the head and other parts of the worm)."
With all these ways C. elegans mirror humans, plus transparent cells easily seen with a microscope, it’s no wonder so many studies have been done on them. Rabinowitch and his team wanted to find out whether reconnecting neurons artificially would restore the worms’ ability to smell after the neuronal pathway connected to that ability was broken. They cut out a pair of neurons to prevent a message fired from one end of the olfactory circuit from getting to the other. This got in the way of their chemosensation, or how they responded to chemical stimuli such as things that they would normally smell to either sense food or keep themselves out of danger.
As predicted, the worms were temporarily unable to smell. The scientists then rerouted the pathway by going into their genes and expressing the protein connexin, which formed an artificial electrical synapse. Synapses are the connections between two cells through which messages are delivered by a neurotransmitter, either from neurons to neurons or neurons to muscle cells. This method connected the neurons which had been at either end of the pair cut out earlier.
"Like any other protein, the connexin protein (from which electrical connections between neurons are constructed) is encoded in DNA," said Rabinowitch. "To produce connexin, a cell has to carry this genetic information and include the correct regulatory elements (some more DNA sequence) that drive cell-specific expression. By inserting the connexin gene sequence together with particular cell-specific regulatory sequences we can cause connexin expression in specific C. elegans neurons of our choice, creating new electrical synapses exclusively between these neurons."
While C. elegans actaully uses innexin for the purpose that we use connexin for, it is similar enough to pass. Rabinowitch was amazingly able to put together the entire DNA sequence he chose and then inject the worms with it just as they egg cells are were forming in their bodies. The cells took on this DNA in their nuclei, which meant his transgene would switch on connexin in the newly hatched worms. Reactivating a circuit like this in a nematode could mean doing the same in humans.
"We can 'mix and match' universal parts between different systems and end up adding new synaptic connections into the nematode nervous system that play the same roles as in the human nervous system, and we may learn from the outcome in nematodes what could be expected someday if a similar approach were to be implemented in humans,” Rabinowitch said.
There has been some success with creating artificial links through computer technology. Brain-computer-brain interfaces (BCIs) can process incoming brain signals and translate them before sending them through the rest of the pathway they started out on. Such devices have helped people with brain damage to regain certain mental faculties or even the use of a limb. Of course, humans are much more complicated than your average nematode, but the similarities between homo sapiens and this unassuming life-form mean the potential is there.
"C. elegans, could help with drawing general principles of synaptic bypass design, comparing between alternatives and learning about their relative effectiveness, studying the impact of new synapses on existing behaviors and other neural circuit functions," siad Rabinowitch. "The next step would be to use this basic knowledge to test the approach in more complex systems, in collaboration with other research groups."
Human brain damage might have not been completely reversed yet, but it’s kind of mind-blowing when you think about what a brainless worm you can barely see is capable of.