Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
A new wearable could detect and reverse drug overdoses in the future
Go, go Gadget...
Opioid-related overdose deaths remain a major public health issue in the United States. More than 800,000 people have died since 1999, according to the CDC, and the yearly numbers are rising. In 2019 alone, opioids accounted for nearly 50,000 deaths, more than 70% of total drug overdose deaths, in the U.S.
An opioid overdose results in decreased respiration and ultimately the cessation of breathing, cardiac arrest, and death, if left untreated. These effects can be rapidly reversed through the administration of naloxone (Narcan) by medical professionals or bystanders. However, many overdoses occur while a person is alone or otherwise accompanied by people who don’t recognize the signs or know how to help in the moment. As such, improved methods of detecting overdoses and administering rapid care can save lives.
A new paper by Justin Chan from the Paul G. Allen School of Computer Science and Engineering at the University of Washington, and colleagues, explores the development and deployment of a novel wearable for use in individuals at risk of opioid overdose. Their findings were published in the journal Scientific Reports.
“Unfortunately, 52% of fatal overdoses occur when a person is alone, and even if they are witnessed by friends or family, they may not be aware an overdose is occurring. What we’ve developed is a wearable injector system that can detect respiration patterns associated with an opioid overdose and automatically administer naloxone to rapidly restore respiration,” Chan told SYFY WIRE.
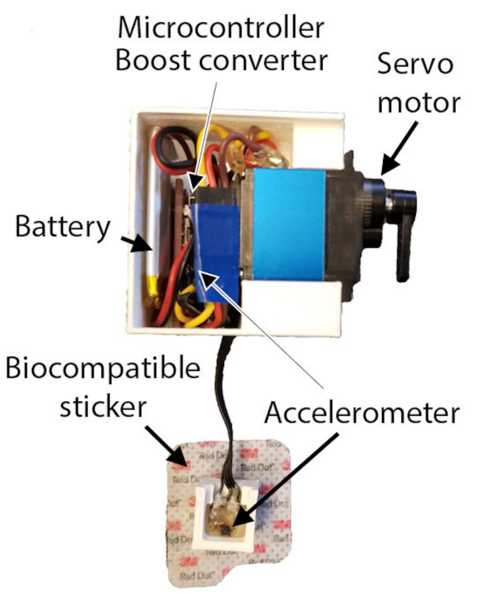
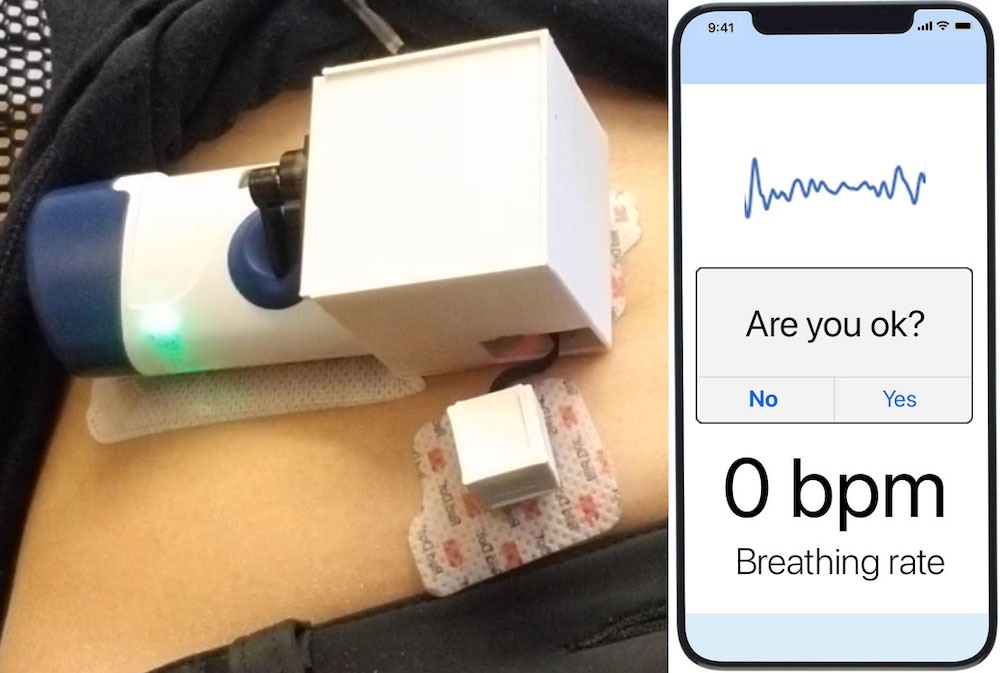
Unlike other more common wearables, the automatic injector doesn’t go on the wrist or anywhere that it is readily visible to onlookers. It’s applied to the stomach where it is hidden from view and has ready access to easily injectable tissues.
“Discreetness and comfort are high priorities for these sorts of devices. Traditionally, naloxone is administered either nasally or through intramuscular injection. So, having something the size of a smart watch that sticks to the abdomen, like a bandage you can take on and off, is the form factor we decided upon. It seems to be acceptable and discreet to people who inject opioids,” Chan said.
In order to establish proof-of-concept, researchers carried out experiments under different settings. They first needed to establish the device’s ability to accurately track respiration rates in opioid users. This was carried out at the InSite Supervised Injection Facility (SIF) in Vancouver. Informed consent was gathered from opioid users at the facility before being fitted with two devices. The first was an existing respiration belt which acted as a reference standard, the second was the sensor developed for the naloxone wearable. Participants were asked to breathe normally for two minutes prior to injection to provide a baseline. Respiration was then observed for five minutes post-injection, as the first five minutes are the time period with the highest risk. Of the two participants, two experienced apneic events without an overdose, and researchers confirmed the sensor correctly detected those events.
The second test took place in hospital setting with healthy participants and confirmed the ability of the device to detect apneic events and deliver medication. “Participants simulated apneic events associated with an overdose, by holding their breath, and our devices successfully identified these events and injected patients with naloxone. We then verified, based on a blood draw, that naloxone was present in their blood,” Chan said.
Naloxone is an opioid receptor antagonist, meaning it binds to the same receptors stopping and reversing the effects of naloxone in the body. Importantly, it is a benign drug and only works in the presence of opioids. There’s no evidence of adverse reactions in healthy people, meaning the study participants weren’t at risk during the study. It also means that wearers of this device are at no apparent risk in the event of a false positive triggering a dose of naloxone from the device.
“We are very interested in making these devices widely available,” Chan said. “That first requires FDA approval. We know the FDA is working to accelerate efforts to address this public health problem and release special guidance for regulation of these types of devices. That’s something we’re looking into.”
A survey of 97 adults with a history of opioid use indicated roughly three quarters would be willing to wear a device like the one designed by Chan and colleagues, and roughly the same percentage stated a willingness to wear the device all or most of the time. Life-saving intervention could be a simple matter of getting these devices into the hands — or onto the abdomens — of the populations who would benefit from them. Though regulatory barriers and social stigma are challenges, researchers are hopeful that someday soon, at-risk individuals will have access to devices capable of knowing when they are in trouble and then helping, even when there’s no one else around.