Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
Solar UV radiation on Earth's surface may have been more intense eons ago than we thought
New models show Earth had less oxygen and therefore less ozone billions of years ago.

About 2.5 billion years ago, the Earth did not look at all like it does today.
For one thing, there was no life on land; what organisms existed at that time lived in the oceans. Also, the atmosphere had very little or no free oxygen in it. But then an immense catastrophe occurred: A kind of blue-green algae called cyanobacteria evolved photosynthesis, the ability to convert sunlight into food. As a byproduct, they created oxygen. This gas, created in massive amounts over a long period of time, polluted the air — at least, to anaerobic life it was poison — and changed Earth forever.
Today our air is about 22% oxygen, but it hasn't always been that way. The amount has fluctuated over time, increasing and decreasing over geologic timescales. It took about 200 million years after the above-described Great Oxygenation Event for atmospheric levels of oxygen to become significant, but even after that the levels fluctuated, sometimes dropping to 10% and even 0.1% of the present levels. Around 500 million years ago levels increased again, and the fluctuations steadied a bit, going from 10% to 150% of today's levels. This roughly corresponds to the time of the Cambrian Explosion, when life underwent a sudden diversification, though its not clear if that's tied to the atmospheric oxygen levels or not.
These numbers are in part based on geological study of minerals from that era, but also models of how the atmosphere behaves. However, these models used a simple one-dimensional approximation (basically, assuming the Earth was covered in layers or air that are the same everywhere at a given altitude, like shells). A new study just published (link to paper) has examined these models using three dimensions, which allows for far more complicated mixing of layers.
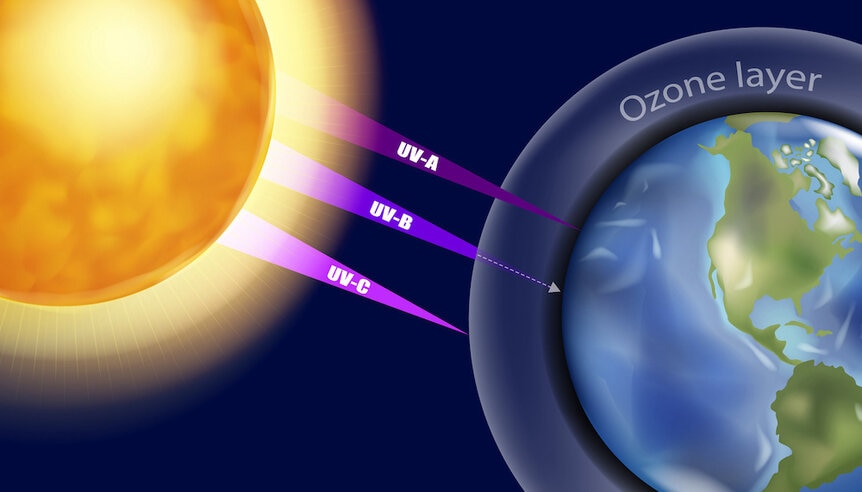
What they found is that the ozone layer, which protects Earth surface from the Sun's damaging ultraviolet radiation, changed by far more in their models than earlier models predicted, and at times the ozone layer was only about 1/5th of what those earlier models showed.
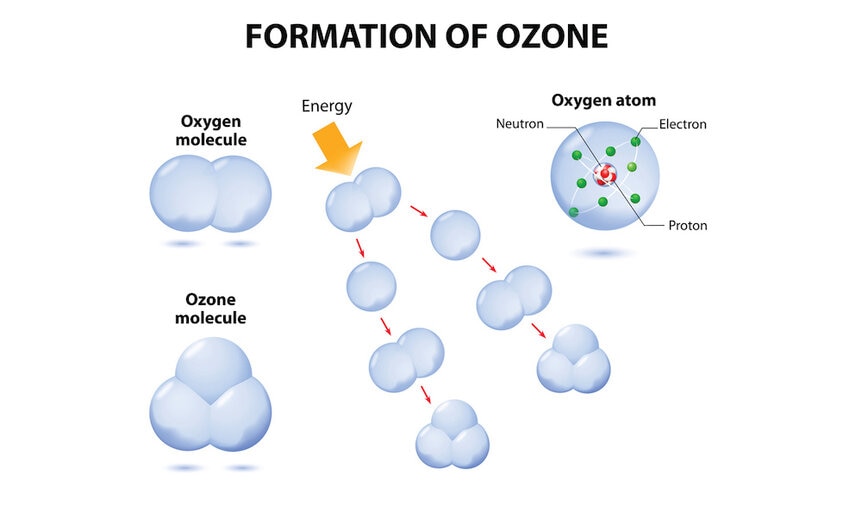
Ozone is a molecule consisting of three oxygen atoms bound together. If it's hit by UV light ozone absorbs it and breaks apart, protecting us on the ground from that UV light. The amount of ozone in the air depends in part on the amount of oxygen in the air — a molecule made of two oxygen atoms bound together — and the amount of ultraviolet light the Sun emits. In this case it's a different wavelength of UV light, called UV-C, that breaks up oxygen in the air which then recombines to make ozone.
As it happens, the Sun was fainter billions of years ago, and the scientists had to accommodate that in their models. The Sun generates energy by fusing hydrogen into helium. That helium builds up in the core of the Sun, gets compressed, and heats up. So, over time as more helium is dumped into the very center of our star, it gets hotter. The rate is very slow, but is significant over billions of years.
Before these models it was assumed that the ozone layer got thick enough to protect Earth's surface once the atmospheric oxygen content was about 1% what it is today, but these new models show that ozone doesn't get built up enough until the oxygen was more like 5–10% of current levels. There were long periods when atmospheric oxygen was lower than this, which means, according to the new models, the ozone layer would've been correspondingly lower.
What does mean for life on the surface?
That's not clear. UV light is very damaging to human skin, for example, and can damage DNA in cells. Water would have protected aquatic life, but once life moved onto land that UV may have added evolutionary pressure to adapt to it. Many bacteria, for example, have greater abilities than we do to repair damaged DNA (D. radiodurans comes to mind), so this may have killed off vast numbers of individuals and species before they adapted to it.
Their three-dimensional modeling also showed that oxygen levels fluctuated differently in different parts of Earth as well, so life may have had a better chance in some places over others, which also would've affected how life took hold on land and how it developed over time.
This new result also affects astrophysics as well. We have ample evidence there was liquid water on Earth billions of years ago, but the Sun at that time was only 70% as luminous as it is now. Naively, one would think the Earth would therefore be frozen; this is called the faint young Sun paradox. It's been thought that perhaps the atmosphere back then was thicker with methane, which is a greenhouse gas and could've warmed the Earth, resolving the paradox. However, the new results imply that methane would've been in short supply, so that's unlikely to be a solution (a recent study may yet save the day, showing the Moon may have heated the Earth in its early days enough to break the paradox).
One-dimensional modeling is simplest, and easier to program into a computer, which is why it's commonly used when first tackling a problem. Adding more dimensions makes modeling much, much harder, as things get complicated rapidly (for example, turbulence in the way fluids flow is an issue that only pops up in higher dimensions, and is incredibly difficult to model). But 3D is also more realistic, so while using it is very hard the results are more likely to accurately reflect reality. Having said that, no doubt scientists will continue to poke and prod and tweak these new models to see what else they can conclude as more realistic conditions are added. It'll be interesting to see if these results stand up, and what new things we can learn about what the Earth was like eons ago.