Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
Pork or beef? Stem cells pave the way for lab-grown meat
Would you eat a burger grown in a lab?
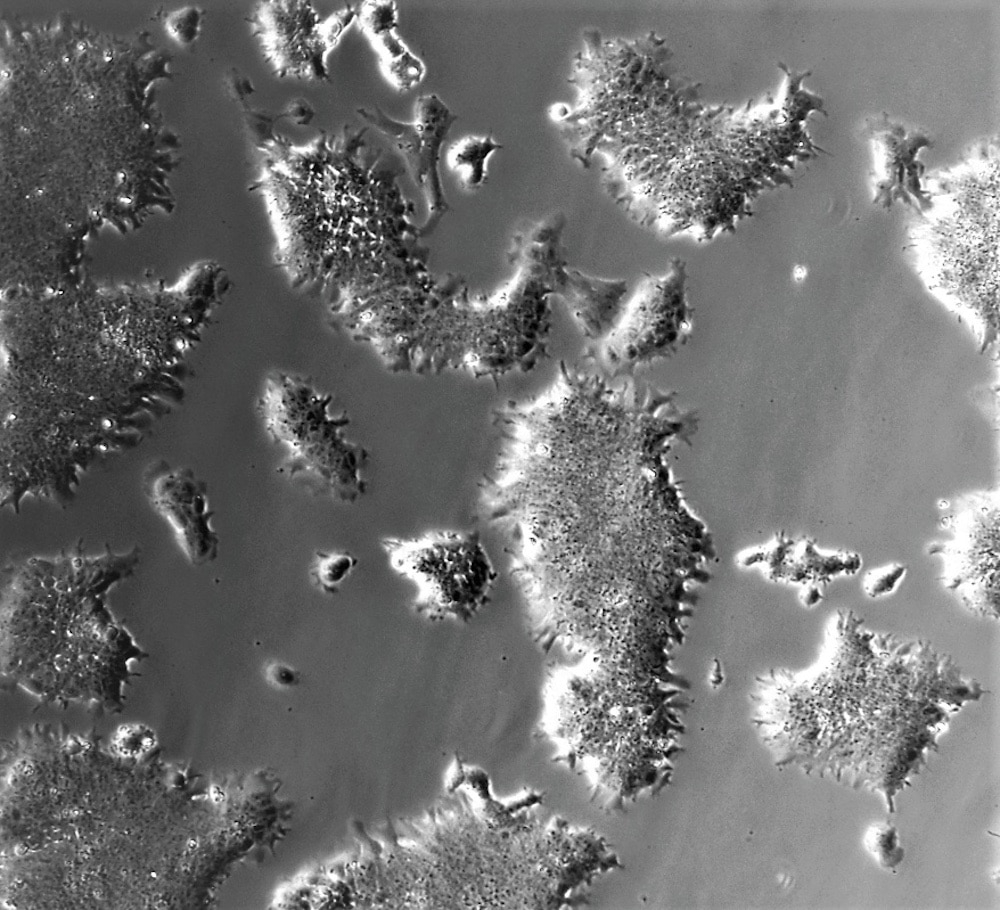
For years, stem cells have been a sort of magic word thrown around as a potential solution for all sorts of challenges in biology and medicine. Certainly, the potential inherent in cells which retain their ability to differentiate into different tissues is nothing to scoff at, but acquiring these cells in a reliable way remains a challenge even decades later.
In order to grow cells in a laboratory setting, researchers rely on additives like fetal calf serum, which is extracted from the coagulated blood drawn from a fetal cow. Once undesired portions of the blood are removed, you’re left with a serum which is pretty good at helping cells to grow. The whole process, however, is messy and difficult to duplicate precisely. As a result, we can’t be certain that cell lines grown in this way are always the same, something which is necessary when reproducing experiments and is especially important if you want to manufacture lab-grown meat, an increasing area of interest. That problem might now be solved, and lab-grown meats might be on their way to your dinner table.
A recent paper by Ramiro Alberio from the School of Biosciences at the University of Nottingham, and colleagues, describes a new well-defined method for stem cell expansion which could open doors to mass manufacturing of animal tissues for human consumption. Their findings were published in the December issue of Stem Cells and Regeneration.
“For industrial applications or food production where regulators require that all components are as defined as possible, current methods have a lot of problems,” Alberio told SYFY WIRE. “Moving to chemically defined conditions is an important prerequisite for making a product that will enter the food chain.”
The team found that only a few key criteria were needed to successfully grow stem cell lines in a lab and, surprisingly, that the same setup was effective in cells from multiple animal species. As a result, one system can grow tissues from cows, pigs, and sheep. In fact, the same criteria also apply to human and mouse cells, providing an added benefit for researchers while we’re all chowing down on synth-steaks.
“We were able to come up with a single unifying recipe for the key components needed by stem cells,” Alberio said.
Because researchers are working with stem cells — as opposed to muscle cells which have already differentiated — they’re able to take a single sample and grow the various tissue types needed to replicate the meat you might find at the grocery store.
Muscle, fat, and connective tissues were all successfully grown in the lab, but this process offers meat with some assembly required. Scientists don’t construct those cells into fully-fledged meat products in the lab. Instead, it would be up to consumer companies to take these cellular building blocks and put them together into something you could fry up at a barbecue.
Like your favorite chain restaurant, it’s important to be able to produce a universal end-user experience free from the guesswork usually associated with stem cell production. Especially as cultured meat products are likely going to be met with some skepticism from the consumer public, scientists want to ensure they’re delivering a product which can be trusted. One of the major benefits of this process is its ability to deliver the same product time after time for at least several years, and probably longer.
“These cells are very stable in culture. We’ve grown them for about three years, and they are still stable. I wouldn’t be able to give a finite number of how many times we can expand these cells though,” Alberio said. “Over time, cells are likely to acquire chromosomal abnormalities simply because the number of mitotic divisions is so huge. Inevitably there will be aberrations that emerge.”
Alberio clarified, however, that those mutations are rare. Over the course of three years, they’ve seen very few, but he urged the importance of closely monitoring the cultures and handling them as well as possible to ensure their stability over time. Should a particular line become unstable, it would likely need to be scrapped and replaced with a new sample. One way to minimize mutations is to grow the cells in a low oxygen environment, which is one of the ways this lab differs from others around the world.
“We use low oxygen for growing the cells. They grow at 5% oxygen rather than 20%. This helps prevent reactive oxygen species being released in the medium which can have a detrimental effect on the chromosomal integrity. Keeping the cells in the best possible conditions allows us to expand the lifespan considerably.”
The unique laboratory setting has other benefits as well, benefits which stand to revolutionize the meat production industry if they’re implemented. Because they are grown in a carefully controlled environment, there’s no need for the introduction of antibiotics which are prevalent in factory farming. Alberio also noted the potential positive impact on climate change. Cows are known for their tendency to release methane into the atmosphere which contributes to warming. Globally, livestock are responsible for 14.5 of all greenhouse gases, a number which could be drastically reduced through cultured meat production.
“When this technology becomes more mature and streamlined, we should be able to produce meat with no methane emissions whatsoever. There would be some CO2 emissions, but methane would be eliminated from the process of meat production,” Alberio said.
The team is now looking to enhance the process by moving from 2D cultures to 3D culture systems which would increase the total volume of tissues they can produce. They’re also looking at producing cell lines from living animals rather than embryos and culturing cells from specific individuals and breeds.
All of which means that menus of the future might include options from particularly famous animals, well-known for their especially delicious tissues, all while they’re happily living their lives somewhere. Win-win for everyone.


























