Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
Shapeshifting robot broke out out of a jail cell by temporarily liquifying
Optimus Prime would be proud.

When I, Robot (now streaming on Peacock!) came out almost two decades ago, the year 2035 still felt like a far-off future filled with possibilities both wonderful and terrifying. Now, not so much. The year, and some of its potentialities, are rapidly approaching. In the movie, humanity has crafted a veritable army of service robots, courtesy of the fictional corporation U.S. Robotics. The robots do everything for us, from running errands to cooking and cleaning, and everyone has welcomed them into their homes and workplaces without so much as a second thought. Everyone, that is, except homicide detective Del Spooner (Will Smith).
Years earlier, Spooner was in a car accident and was saved by a robot. You’d think that might ingratiate him toward them, but quite the opposite. That same robot allowed a young girl in another car to die, and Spooner has never been able to square his own survival with the girl's death. A human might have made a different choice, but robots are bound by their programing. It’s a cold calculus and sometimes we might not like the results.
Fortunately, we haven’t yet had to face up to the inevitable consequences of everyday interactions with increasingly intelligent machines, but that day is quickly approaching. Recently, new artificial intelligences from image generators like DALL-E and text generators like ChatGPT have reignited the public conversation about what artificial intelligence is going to mean for us and how the world will change in its presence. Now, scientists have demonstrated a tunable metal material which might allow the next class of robots to change their shapes at will.
RELATED: M3GAN, HAL, Skynet, and more: 10 time A.I. tried to kill us in movies
Carmel Majidi, from the Soft Machines Lab at Carnegie Mellon, and colleagues, have demonstrated the potential of low-melting-point metals for robotics and medicinal applications. The work was recently published in the journal Matter.
Their material is relatively simple, comprised of only two real ingredients: gallium and magnetic particles. When we think of metal we’re often thinking of iron, which has a melting point of 2,800 degrees Fahrenheit (1,538 Celsius). Those aren’t temperatures we typically encounter, so we think of metal as being hard and rigid. Gallium laughs in the face of our expectations by having an incredibly low melting point. It melts at 85 degrees Fahrenheit (30 Celsius) which is colder than the human body. Just picking it up is enough to send it oozing through your fingers. But researchers didn’t use direct heat to manipulate their material. They used something a lot weirder.
“We apply this alternating magnetic field that induces current within the gallium and that current heats it up. That’s how we were able to elevate the temperature. What’s nice is it’s totally wireless and can penetrate through surfaces, so it doesn’t require direct line of sight,” Majidi told SYFY WIRE.
While traditional engineering has relied on increasingly complex combinations of mechanisms, scientists and engineers are moving into an era of simplification. The goal now is to achieve functionality not through mechanical force but through innate properties of your chosen material. Metals with low melting points are of particular interest because they offer stiffness tuning and electromagnetic properties which can be exploited to change their phase, their shape, and move them around. That’s something nature achieves with relative ease, but which our machines struggle to duplicate.
RELATED: I won’t let anything harm you: The science behind 'M3GAN' and lifelike robots
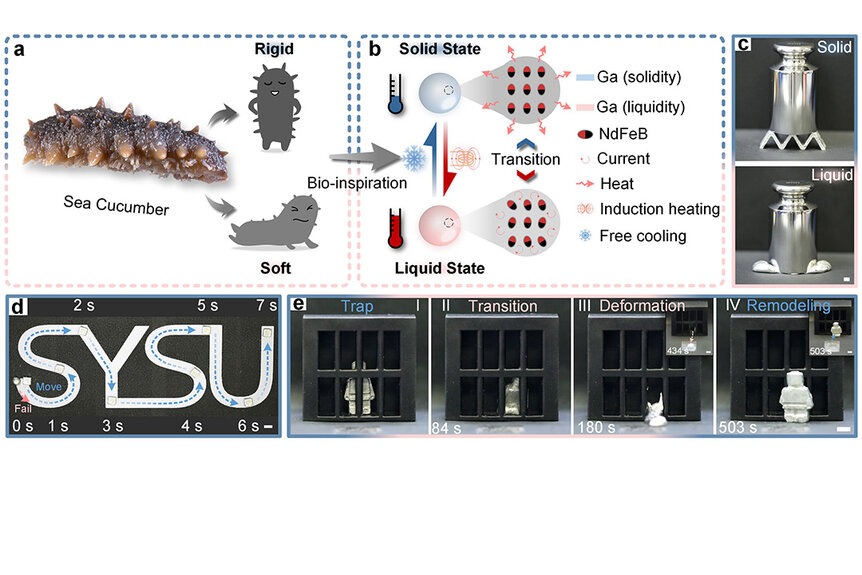
“This work was inspired by the sea cucumber, this marine organism that can pretty dramatically change its stiffness. It goes from being pretty rigid and solid to almost gel-like and fluidic. It’s an important adaptive mechanism they use to avoid predators. They can maneuver themselves into really fine spaces and hide away. It was that combination of stiffness and shape-changing we were trying to mimic,” Majidi said.
And mimic it they did. During a demonstration of their material, researchers crafted a small figurine and placed it inside a proportionally small jail cell. Then, using only magnetic fields, they increased the temperature of the metal, transforming it onto a liquid blob and maneuvering it out of the cell (pictured below). Changing back into a solid just requires a drop in temperature, but getting back into the shape of a figurine requires a manual molding process. At least for now.
“It was meant to convey a vision. What’s being demonstrated is the ability to take a figurine and melt it down using that electromagnetic effect. Then use magnets to move that liquified blog out of a confined space. That’s what the demonstration shows,” Majidi said.
It’s a fun proof of concept, but the likely first application of these materials isn’t in shape-changing robots, it’s in medicine. Getting there will require making some changes to the material. Because gallium’s melting point is well below the average human body temperature, we couldn’t induce the state change from solid to liquid and back again. That’s why researchers are experimenting with gallium alloys. By combining gallium with other non-toxic metals like bismuth, they might be able to elevate the melting point to within the right temperature range. The idea is to achieve a material that could undergo the same phase change, from solid to liquid and back again, inside the human body.
More research is needed to ensure that those alloys are safe for direct contact with cells. Experiments so far have suggested they are safe in specific use cases but can impede cell growth. That’s something we’ll have to contend with before you can get shape-changing robot meds over the counter. It might be a matter of finding the right combination of ingredients.
RELATED: San Francisco police propose allowing robots to use deadly force against people
In the meantime, researchers have already tested the material’s ability to retrieve objects or deliver medicines. In one example, a solid piece of material is placed inside a model stomach. When the magnetic field is applied, it changes to a liquid and releases a medicinal compound contained inside. In another example, a solid piece of material moves toward a foreign object, liquifies and surrounds it, and solidifies again before extracting itself from the body. The future of these materials remains to be determined, but their potential is significant.
“What gets me excited about this work is that it really demonstrates one more way metals like gallium that have a very low melting point can endow systems with really extraordinary properties that we’ve never seen before in conventional engineering systems,” Majidi said. “I think there’s a lot more to discover about these metals and my hope is that they become much more common in engineered systems, whether it’s medicine, electronics, robotics, or whatever. There’s a lot more to explore.
Who needs to change shapes when you have an army of robots at your disposal? See how robots try to take over the world the old fashioned way in I, Robot, streaming now on Peacock.