Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
DNA polymers will give soft robots good genes
They're going to inherit what metal robots wish they could.

In the not-so-distant future, soft robots could be doing everything from exploring the darkest depths of the ocean to scoping out lunar and Martian lava tubes — but they can break down.
Swarms of bots which could potentially carry out those tasks will be made of flexible polymers that allow them to move in ways that are impossible for a clunky metal robot. However, extreme environments are punishing for even the toughest materials, so you need to come up with something that can survive deep in the Earth or out in space. Turns out all we need to do is pass our genes onto the soft robots that could soon be navigating alien territory.
Wait. How exactly can you pass on genes to a thing that isn’t even alive? You sort of can if you use DNA strands to link polymer molecules, since the bonds that hold together those strands have the predictable behavior that polymers need to survive. Researcher Xiang Li of Hokkaido University in Japan, who led a study recently published in Advanced Materials, simulated different DNA sequences and created a gel polymer that uses those properties to its advantage.
“We focused on the adjustable mechanical behavior of double-stranded DNA,” he told SYFY WIRE. “To effectively transfer the local mechanical event in the DNA links to the entire gel behavior, we need to make the gel homogeneous.”
Previous research teams had attempted to make the DNA structure work, but something was missing. Li thinks they did not focus on the potential for these structures to adapt (and therefore allow the gel to adapt to its surroundings) enough. Bonding polymer molecules in the shape of DNA strands gives a gel enormous potential. Those molecular structures are sensitive to temperature, biocompatible, and soluble in water, which means that besides crawling into the unknown in the form of soft robots, they could also help revolutionize medical treatments.
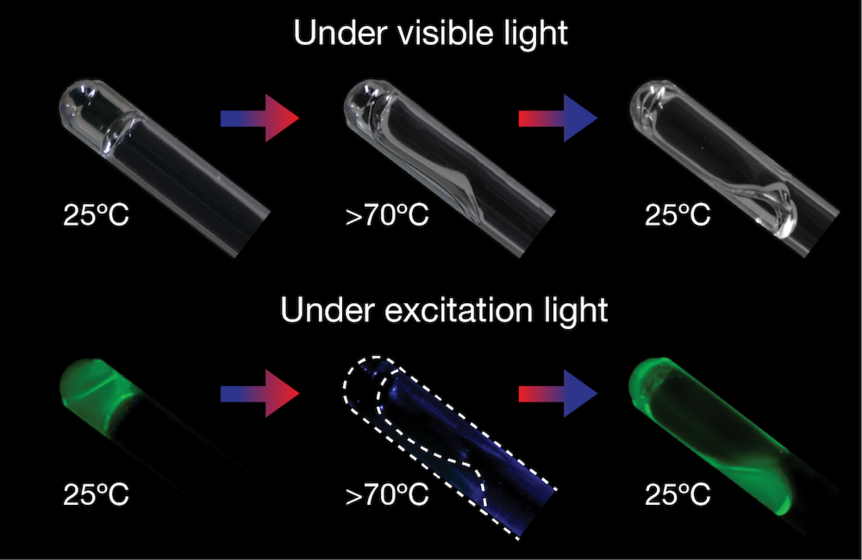
Such gels also need to be homogeneous, or the same throughout, because the effects of mechanical actions are transferred throughout them. Li and his team took their strategy for making homogeneous gels and applied it to using DNA for linking polymer molecules. The double helix will only form without being disrupted, which is is why it is so important for a gel to be homogeneous and not heterogeneous. After software simulations told them which DNA sequence was most likely to hold up, they used that sequence on molecules of polyethylene glycol (PEG).
“The designed sequences will only form the double helix structure without unfavorable structures,” said Li. “In addition, this pair of DNA sequences exhibit a clear two-state transition from the double to single-stranded DNA, making the gel's mechanical responses very simple.”
What the team had to do next was link connected PEG molecules with complementary DNA sequences, and to do that, both DNA strands and PEG needed to be dissolved on their own before anything could happen. They were then combined in a test tube which was in a hot water bath. For the gel to form, the temperature of the water surrounding the test tube was gradually cooled to room temperature. Turned out the simulations had led the team on to the right DNA sequences. The gel was elastic and able to repair itself as they had predicted.
Because DNA strands linked the PEG molecules homogeneously, the gel stayed together unless it was exposed to temperatures over about 145 degrees Fahrenheit, which would break down the bonds. Materials like this gel are so adaptable, they may soon be assisting tissue regeneration methods that need materials to boost the growth and differentiation of cells as much as possible. This could lead to entire organs being created in vitro further out in the future. DNA gels will also be amazing for soft robots, since they can adapt to almost anything.
“As we can flexibly tune the mechanical responses of the DNA gels by simply altering the DNA sequences, we believe DNA gels would contribute to soft robotics,” said Li.
You could say the abilities of the next generation of robots are in their genes.