Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
Dr. Frankenstein can take a day off, because these micro-hearts create themselves and beat on their own
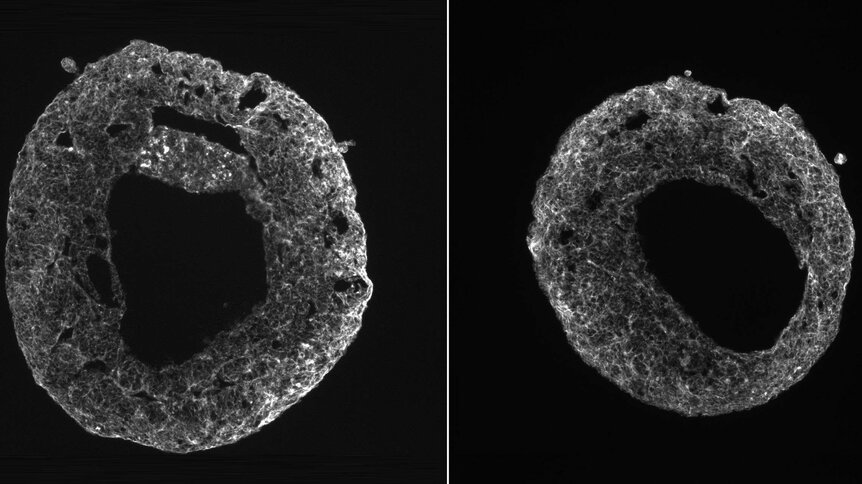
It sounds like Mary Shelley having a fever dream. Human hearts, beating on their own, emerged out of seemingly nowhere.
At least it would appear as if those hearts (which were really just the size of sesame seeds) materialized in petri dishes because they self-organized out of microscopic stem cells. They Frankensteined themselves after biologist Sasha Mendjan of the Austrian Academy of Sciences figured out how to switch on the signaling pathways that activated the cells into organizing themselves into all the right structures. After a week, these organoids or “cardioids” were contracting to squeeze fluid inside a cavity. They had eerily started to beat.
“The work is important for us because a heart chamber is the basic unit of the heart, and we managed to grow it naturally via developmental mechanisms, not by engineering and forcing the cells to do something,” Mendjan tells SYFY WIRE. “This ensures also a physiological response we see when we try to mimic diseases.”
Self-organizing organoids, including the brain and even the labyrinthine intestine, have been created for every major organ in the human body except the heart, which has proven exceptionally difficult until now. The problem with engineering organoids is that you have to build the structures out of cells in an unnatural way — something Dr. Frankenstein probably would have done. Allowing cells to create an organoid on their own gives scientists a better idea of what developmental phase congenital diseases would start to form. It also allows scientists to target the early stages of cancer or cardiac arrest.
Mendjan, who led a study recently published in Cell, realized what other researchers had been missing when he and his team tried to push pluripotent (specialized to form certain structures) stem cells into creating a heart. The answer was hidden in signaling pathways. These are the result of molecules in a cell producing chemical reactions that control a certain function within that cell. There are six of these that are known to contribute to the embryonic growth of the heart, which is the first organ to function in utero. Previous studies had assumed that not all six were needed. In this situation, it turned out that they were.
“We activated signaling by adding proteins or small molecules to the media, which either activate or inhibit signaling,” Mendjan says. “The pathways needed are known to be important, but it had been unclear exactly why and at what stage until we separated the pathways required for specification of cells versus those that change their shape.”
Another thing the team wanted to demystify is whether these cardioids would take action if damage was inflicted on the tissue. After freezing tissue with cold steel, they noticed that cardiac fibroblasts, which are cells that produce connective tissue to start healing wounds, immediately speed towards the damaged tissue in an effort to save it with proteins. Proving that these cardioids are robust enough to undergo testing like this will make pharmaceutical companies less hesitant to contribute medications to such research. Recreating everything from congenital defects to cardiac arrest, so treatments can be tested in vitro, could eventually save lives.
Now that he and his team have the signaling pathways for self-organizing cardioids down, Mendjen plans to continue the research to make them stronger and more complex so they can model causes of disease on an actual human heart. The heart has four chambers. He and his team want to keep working with signaling paths to activate whatever creates the extra chambers that are responsible for some congenital defects.
“The future is going towards multiple chambers, so the model needs more complexity as well as a connection to a circulatory system,” he says. “Our challenge is to combine complexity with control and robustness, so the model can be used for purposes such as testing treatments.”
Mary Shelley would have been inspired, because this is exactly the type of thing that can fuel a heart-pounding horror novel.