Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
The building blocks for RNA-based life have been found… in the center of the Milky Way
Did life here begin out there? Or maybe its complex ingredients?

We don’t know exactly how life arose on Earth.
For one thing it was a long time ago: Roughly 3.8 billion years in the past, give or take, and records of anything that happened from that period in Earth’s very ancient history are spotty. For another, we don’t know the chemical path life took. Surely simple molecules built up into more complex ones, eventually becoming able to store information and self-replicate. And then, abracadabra, DNA popped up and the rest is biological history.
But those are big, big steps, and it’s unclear how they happened. One idea is called RNA World. It posits that RNA — ribonucleic acid, similar to but simpler than DNA — was the basis for life on Earth for a while, before the development of proteins and DNA, which together make up the big molecules necessary for life on Earth today.
But how did RNA arise? Simpler molecules like ribonucleotides had to develop first, because they are the building blocks of RNA. And they come from even simpler molecules, among them a kind called nitriles. These have a carbon atom strongly bonded to a nitrogen atom, and the pair is then bonded to some other element or molecule.
Ok, fine. So where did nitriles come from?
Here’s the fun bit: Maybe space.
It’s possible that all these chemicals arose natively on Earth as the crust cooled and gases poured out of volcanic vents and such, chemically enhancing the surface. However, we’ve also found some pretty complex molecules in meteorites, including amino acids that are necessary to make proteins. That means that some of these complex biological precursor molecules can be made in space.
These meteorites formed in the solar nebula, when the solar system was just a cloud of gas and dust collapsing down to form the Sun and planets and other assorted rocky and icy bits. That in turn means these molecules may have formed in cold nebulae called molecular clouds. These can be gigantic, hundreds of light years across, like the Perseus Molecular Cloud and the Orion Molecular Cloud.
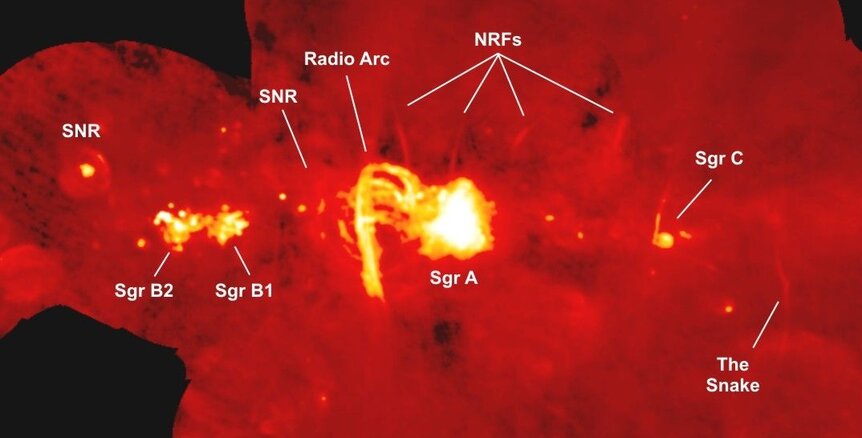
Another big molecular complex is called Sgr B2, very near the center of the Milky Way. In it is a cloud called GCM0.693-0.027, which is well suited to look for complex molecules. In fact, nitriles have been found in it before, but a new study turned to a pair of telescopes to look for more. Using the IRAM 30-meter and Yebes 40-meter radio telescopes in Spain, astronomers detected quite a few more of these molecules in the cloud, including ones with such fun names as cyanic acid, cyanoallene, and cyanopropyne [link to paper]. They also made tentative detections of a couple more, but these cannot be confirmed as yet.
They used radio telescopes because of the odd way these molecules emit light. We see gas in space because the elements in it absorb light and respond by glowing at very specific wavelengths — colors — that act like fingerprints in IDing the elements. Hydrogen emits light at a different set of colors than, say oxygen, because the electrons in hydrogen atoms jump from different energy levels than oxygen atom electrons do.
Complex molecules can do this as well, but the energy levels of the electrons overlap so much that the light emitted this way isn’t terribly discrete; they absorb and emit energy in bands, wide ranges of energies. However, these complex molecules can rotate, and they do so at specific, discrete energies. It takes an exact amount of energy to get them rotating that way, and to change rotations they emit that exact amount of energy in the form of light. These are called rotational transitions, and in general the light emitted is in the radio part of the electromagnetic spectrum. Hence the use of radio telescopes to detect them.
These new observations add to the list of these types of molecules detected in space. Some of them have been found in the Taurus Molecular Cloud as well, which attests to their ubiquity; they seem to form easily in different clouds.
That’s important. The Sun and planets formed from a cloud that may have once been part of a giant molecular cloud, now likely long since gone all these eons later. But if these nitriles and other complex molecules formed in our precursor cloud as they did in others, they could have condensed onto icy bodies that coalesced from that material farther out from the Sun than the orbit of Neptune.
There’s an idea called the Late Heavy Bombardment, where the inner planets of the solar system, including Earth, were pounded by huge impacts from outer solar system objects — they may have been perturbed from their orbits by the giant planets moving around, or some other cosmic event.
Lab experiments have shown that some complex molecules buried in comet-like objects such as these can survive slamming into a planet.
See where is this headed? We see these molecules in molecular clouds, this stuff condenses to form objects in the outer solar system, these objects get tossed inward where they hit Earth, and our planet gets loaded up with nitriles and amino acids and other such fun molecules, basically a home delivery system of ingredients needed to bake up biology.
It’s not proven by any means, but the steps are logical and we do see evidence of them.
Maybe the original Battlestar Galactica was right all along. Life here began out there.
Or at least its precursors did. And that may have been enough.