Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
Jupiter's moon Europa may glow due to salty irradiation

If you're curious about life elsewhere in the solar system then sure, Venus (for better or for worse) and Mars have been making headlines.
But perhaps the best place to look has been and still is Europa, Jupiter's icy moon. We've known for decades it has a liquid water ocean under its surface, and the core of the moon may be rocky and hot, which could supply nutrients to any swimming alien beasties there via hydrothermal vents.
It's also possible that there are interesting chemicals on the surface, which could get transported down into the water below via cracks. The problem is figuring out what's there. Not long ago, evidence of sodium chloride — table salt — was found there; remember life here started in Earth's oceans, which are salty. So that's interesting. Magnesium salt (epsomite) may have been found, too.
But other species of salty molecules have been difficult to pin down. They're featureless in many wavelengths observed, making them difficult to detect. But new research may change that: Scientists found that ice glows when bombarded by radiation, and different salts in it change the glow in characteristic ways, making them potentially identifiable.
This could be extremely helpful! Jupiter's magnetic field bombards Europa with powerful radiation. Very powerful; if you stood outside unprotected on the surface of the moon it would give you a lethal dose in minutes (though freezing to death and suffocating may be more proximate issues). Subatomic particles like electrons and protons get trapped in Jupiter's fierce magnetic field. The planet spins rapidly (a day on Jupiter is about 10 hours), so the magnetic field moves around Jupiter faster than Europa does, slamming those particles at very high energies into the backside of the moon (what we call the trailing hemisphere), the side facing away from the direction it orbits.
If there's salty ice there, would that radiation injection cause it to glow and help us find it?
To find out, the scientists cooled both pure water ice and ice with various salts in it down to Europa-like temperatures (-170°C, or -280°F), then slammed it with high-energy electrons like what you'd get at Europa's surface. As they expected, the ice glowed, but when they changed the composition, what they found shocked them.
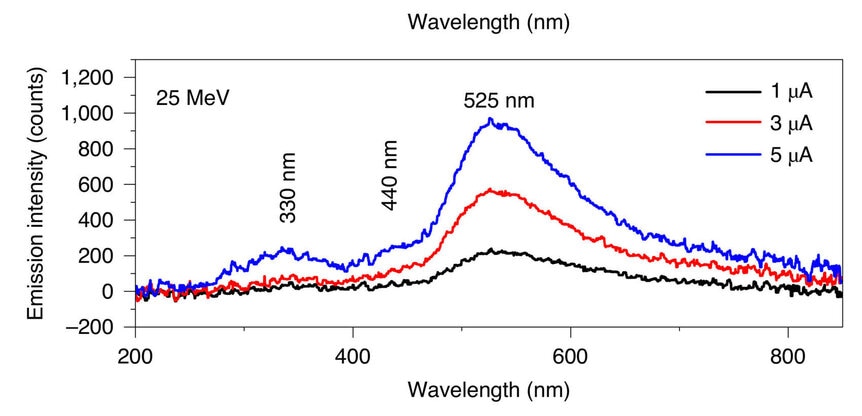
The spectrum (that is, the light from the ice broken down by color) of the pure ice had three prominent features: A bump at a wavelength 330 nanometers (in the near ultraviolet), a "shoulder" (like the edge of a bump) at 440 nm (blue), and a big, broad bump that peaks at 525 nm (green). These are all likely due to water and its constituents (H2O, hydroxode or OH, and oxygen). That overall pattern persisted no matter what salts they used, but the sizes of the bumps changed significantly.
For example, adding table salt dropped the big 525 nm peak quite a bit, but the 330 peak only a little. Sodium carbonate (Na2CO3) nearly erases the big peak!
In other words, the shape of the overall spectrum changes in measurable ways depending on the chemicals in the ice.
This has never been seen before in this way. Previous studies looked at, for example, short-term radiation in ice (zapping it briefly), but in this new one they kept the beam on for long periods of time, to imitate actual conditions on Europa.
This is very interesting. Why? Because in a few years, NASA will be launching the Europa Clipper, a mission to Jupiter that will orbit the giant planet and make many very close flybys of Europa. It's equipped with a camera that has several color filters on it, and, according to these new data, should be able to get enough information on the actual ice spectrum to get a handle on what salts may be locked up there.
The scientists even simulated what they'd expect to see using the Clipper's Wide Angle Camera on its Europa Imaging System instrument, and found that the different colors of the salty ices could be detectable during passes over Europa's trailing hemisphere. It would have to be on the night side, so the Sun doesn't interfere (and likely the geometry will have to be carefully chosen to exclude Jupiter in the Europan sky as well, since that great mucking planet in the sky would illuminate things too), but many such passes should be possible.
They note that observations from the ground and using Hubble haven't seen it, but it's pretty faint unless you're right there. In fact, if you were in orbit around the moon, you might be able to see it by eye. So, depending on the salts there, you might see the night side of Europa glowing faintly blue, green, or teal. How lovely!
If the salts are there, and if they glow, that will tell scientists what sort of chemistry could be going on both on the surface of Europa and inside its ocean, forever hidden away under a thick shell of ice. Could there be alien fishies swimming there?
We don't know, but we're getting closer all the time to finding out.