Create a free profile to get unlimited access to exclusive videos, sweepstakes, and more!
Scientists Have Taken an X-ray of a Single Atom
X-rays let us see deeper inside of atoms, too.

If there’s one thing Quantum Leap’s Dr. Ben Song (Raymond Lee) knows for certain, it’s that the quantum realm is difficult to understand and even harder to predict. Once you drill down to atomic scales, things get very weird, very fast. Individual atoms exist at the fringes of our detection abilities, because of their incredibly small size but also because the energies they exert aren’t in the frequencies we can see.
Even our most powerful microscopes, capable of resolving individual atoms in an image, can’t tell you what those atoms are. At least, they couldn’t until now. Researchers from the Argonne National Laboratory recently developed a tool capable of scanning and identifying individual atoms, for the very first time. The results were published in the journal Nature.
Measuring and Identifying Individual Atoms with X-rays
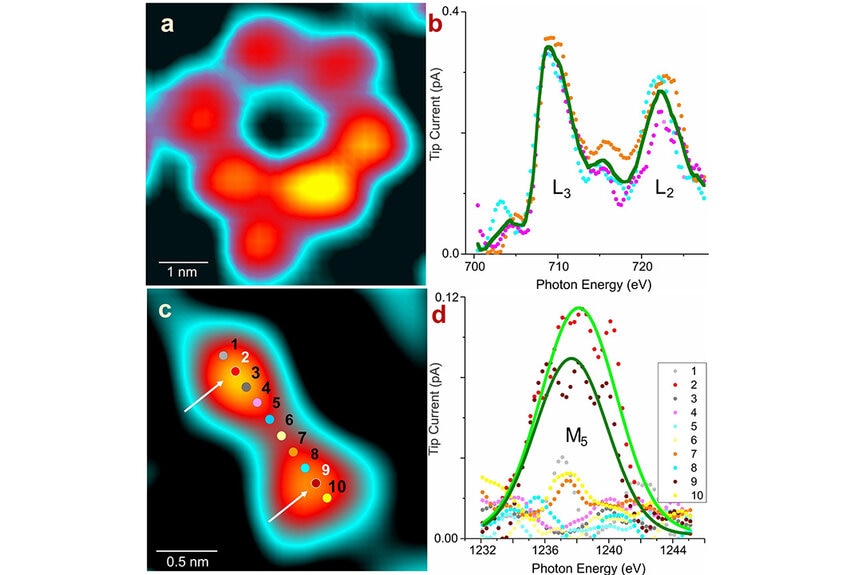
Imaging individual atoms is nothing new. We’ve been taking pictures of single atoms or arrangements of atoms for years, but with one key limitation. Scientists were able to resolve the presence of an atom, but they couldn’t tell you what that atom was unless the sample had been identified in advance.
RELATED: Quantum Leap writers created a ‘real’ time travel cheat code equation for season finale
“We could use atomically sharp needles to resolve the shape of an atom if we scan it very close to the sample surface, using tunneling current or atomic force. In both cases, we can see the atoms, but we cannot tell the type. If you give me an unknown sample, I can give you an image and you can see the atoms, but if you ask me what kind they are, I’d say, ‘You tell me,’” Saw-Wai Hla, a researcher from the Nanoscience and Technology Division at Argonne National Laboratory and author of the new study, told SYFY WIRE.
That’s because existing methods of measuring individual atoms rely on information from the atom’s outer electron shells, and they don’t contain identifying information. To get to the meat of an atom, you need to get to the information encoded in the core level shells. To drill that deep, scientists needed something stronger.
“You need X-rays because you have to excite the core electrons. We shine a synchrotron X-ray at the tip sample junction to excite core level electrons. Now, we can not only see them, but we can also get information about the atom. What is its element? What is its chemical state?” Hla said.
RELATED: Scattered X-rays From Space: Turning Noise Into Signal
When X-rays hit the sample, they add energy to the atom, exciting those core level electrons. Once the internal energy reaches a critical point, a photon is absorbed by the atom and an excited electron is released. By measuring the energy level of the excited electron released, scientists can determine what type of atom they are sampling. “Once we know the energy spectrum, we know exactly which atom is there,” Hla said.
As the technology matures, they may be able to get additional information, such as an individual atom’s magnetic properties. As it stands today, there are already several practical applications which could be implemented using the technology. Researchers suggest it might be used to detect heavy metals or other potentially toxic elements in a water sample or in specific environments like airports or medical settings by passing the detector over a large sample.
Because it is capable of detecting the presence of individual atoms in a sample, it might be used to detect dangerous elements at the lowest possible concentrations. “If you can detect one atom, you can easily detect ten atoms, ten times easier,” Hla said.
In addition to detecting the presence of atoms in existing samples, Hla suggests there are also potential applications in materials science and quantum computing. Maybe if the folks at the Quantum Leap project had this kind of atomic resolution, they might be able to get Ben back home before something catastrophic happens. We can hope.
Catch the original Quantum Leap and NBC’s sequel series, streaming now on Peacock!